MC1000 8通道藻类培养与在线监测系统
MC1000 8通道藻类培养与在线监测系统由8个100ml藻类培养试管、水浴控温系统、LEDs光源控制系统及光密度和溶解氧(选配)在线监测系统等组成,可用于藻类培养与控制实验、梯度对比实验等,适于水体生态毒理学研究检测、藻类生理生态研究、水生态研究等,其主要功能特点如下:

1.8通道藻类培养,每个藻类培养试管可培养85ml藻液
2.LEDs光源,可对每个培养试管独立调节控制和设置光强度和时间,如昼夜变化等
3.光密度在线监测,包括OD680、OD720,监测数据自动存储
4.溶解氧在线监测(备选)以测量分析藻类光合作用等
5.温度、光照控制可用户设置不同的程序模式
6.气泡混匀:可通过调节阀手动调节气流量以对培养试管内的藻类进行混匀
7.可选配O2/CO2监测系统,在线监测藻类光合放氧和CO2吸收
8.可选配藻类荧光测量模块
应用领域:
★多通道同步藻类培养
★同步梯度胁迫实验
★培养条件优化
★控制培养条件与藻类生长动力学监测
仪器型号:
MC 1000-OD:8个通道光源颜色相同,标配冷白光LED
MC 1000-OD-WW:8个通道光源颜色相同,标配暖白光LED
MC 1000-OD-MULTI:8个通道光源颜色不同,分别为1)紫光405nm,2)品蓝光450nm,3)蓝光470nm,4)暖白光,5)绿光540nm,6)黄橙光590nm,7)深红光660nm,8)远红光730nm
MC 1000-OD-MIX:每个通道可配备8种不同颜色的LED光源,LED颜色为1)紫光405nm,2)品蓝光450nm,3)蓝光475nm,4)2个暖白光LED,5)绿光530nm,6)橙红光615nm,7)深红光660nm,8)远红光730nm
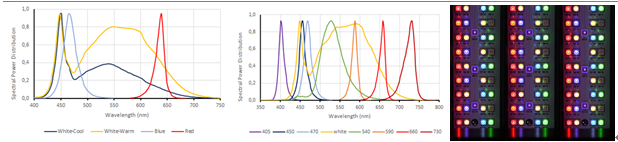
技术指标:
1.藻类同步培养通道:8个
2.培养管容量:100ml,建议培养容量85ml
3.在线即时监测参数:分别监测每个培养管的OD680和OD720,数据自动保存到主机内存中,PIN光电二极管检测器,665-750nm带通滤波器
4.精确控温范围:标准配置高于环境温度5-10℃(与光强有关)~60℃,可选配15℃-60℃(环境温度20℃,需加配制冷单元)
5.加热系统:150W筒形加热器,水浴控温
6.水浴体积:5L
7.水浴自动补水模块(选配):水浴水位因蒸发降低后可自动补水
8.光源系统:全LED光源,可在0-范围内调控,每个通道的光强可分别独立调控
1)MC 1000-OD:标配冷白光LED,可选配暖白光、红光(635nm)或蓝光(470nm)LED;光强0-1000μmol/m2/s可调,可升级至0-2500μmol/m2/s
2)MC 1000-OD-WW:标配暖白光LED,光强0-1000μmol/m2/s可调,更高光强可定制
3)MC 1000-OD-MULTI:8个通道光源颜色不同,分别为紫光405nm,品蓝光450nm,蓝光470nm,暖白光,绿光540nm,黄橙光590nm,深红光660nm,远红光730nm;光强0-1000μmol/m2/s可调
4)MC 1000-OD-MIX:每个通道可配备8种不同颜色的LED光源,光强可达2500μmol/m2/s

9.控光模式:可静态或动态设置光照程序,如正弦、昼夜节律、脉冲等
10.控制单元显示屏:可调控培养程序和显示数据
11.气流调控:通过多管调节阀对8个培养管手动独立调控气体流量
12.OD测量程序:将主机内存中的OD数据下载到电脑中并以图表形式显示,数据可导出为TXT或Excel文件
13.MC实时在线监测分析模块(含专用工作站和软件基础版或版,选配)
1)同时控制2台MC1000(基础版)或无限台MC1000(版)
2)通过PBR软件动态调控光照和温度模式
3)通过光密度(OD680、OD720)变化实时监测藻类生物量
4)对生长速率进行实时回归分析
5)多数据管理功能(过滤、查找、多重导出)
6)可将测量数据、培养程序和其他信息保存到数据库中
7)通过GUI图形用户界面设置培养程序并在线显示测量数据图
8)数据可导出为CSV文件
9)支持GMS高精度气体混合系统(版)
10)用户自编程培养程序(版)
11)设定实验起始时间(版)
12)电子邮件通知(版)
14.GMS150高精度气体混合系统(选配):可控制气体流速和成分,标配为控制氮气/空气和二氧化碳,气源需用户自备
15.恒浊控制模块(选配):带有8个控制阀,可独立控制8个培养管的浊度,由软件自动控制
16.O2/CO2监测系统(选配):8通道续批式监测藻类CO2吸收或光合放氧通量:
1)氧气分析测量:氧气测量范围0-100%,分辨率0.0001%,精确度优于0.1%,温度、压力补偿,数码过滤(噪音)0-50秒可调,具两行文字数字LCD背光显示屏,可同时显示氧气含量和气压
2)二氧化碳分析测量:双波长非色散红外技术,测量范围0-5%或0-15%两级选择(双程),分辨率优于0.0001%或1ppm(可达0.1ppm),精确度1%,通过软件温度补偿,具两行文字数字LCD背光显示屏,可同时显示CO2含量和气压,具数码过滤(噪音)功能
3)气体抽样与气路切换:具备隔膜泵、气流控制针阀和精密流量计,气路自动定时切换功能
17.藻类荧光测量模块(选配):用于测量藻类荧光参数以反映藻类生理状态及浓度,荧光测量程序包括Ft,QY,OJIP-test,NPQ、光响应曲线等,可选配探头式测量或试管式测量:
1)探头式测量:具备光纤测量探头,可插入培养液中原位测量藻类荧光参数
2)试管式测量:具备测量杯,可取样精确测量藻类荧光参数及光密度值
18.通讯方式:USB
19.尺寸:71×33×21 cm
20.重量:13kg
21.供电:110-240V
应用案例:
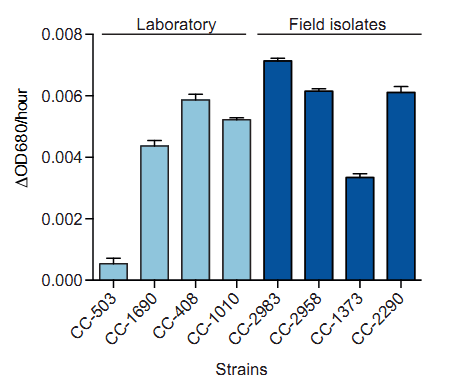
莱茵衣藻全基因组重测序的样品预培养与生长动态监测(Flowers, 2015, Plant Cell)
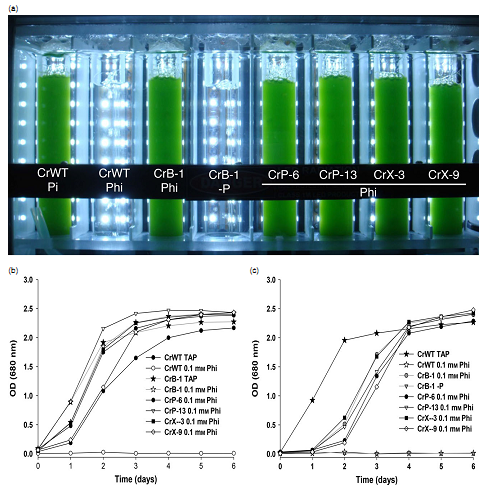
通过基因工程改造莱茵衣藻控制生物污染(Loera-Quezada, 2016, PlantBiotechnology Journal)
产地:捷克
参考文献:
1.Barera S, et al. 2021. Effect of lhcsrgene dosage on oxidative stressand light use efficiency by Chlamydomonasreinhardtiicultures. Journal of Biotechnology 328: 0168-1656.
2.Pivato M, et al. 2021. Heterologous expression ofcyanobacterial Orange Carotenoid Protein (OCP2) as a soluble carrier ofketocarotenoids in Chlamydomonasreinhardtii. Algal Research 55(16):102255.
3.Gachelin M, et al. 2021. Enhancing PUFA-rich polarlipids in Tisochrysis luteausingadaptive laboratory evolution (ALE) with oscillating thermal stress. AppliedMicrobiology and Biotechnology 105: 301-312.
4.Chen H, et al. 2021. A Novel Mode ofPhotoprotection Mediated by a Cysteine Residue in the Chlorophyll Protein IsiA.mBio 12(1).
5.Cecchin M, et al. 2021. CO2supplymodulates lipid remodelling, photosynthetic and respiratory activities in Chlorellaspecies 18(2): 431842.
6.Dixit RB, et al. 2021. Secretomics: A PossibleBiochemical Foot Printing Tool in Developing Microalgal Cultivation Strategies.doi: 10.21203/rs.3.rs-163118/v1
7.Kareya MS, et al. 2020. Photosynthetic CarbonPartitioning and Metabolic Regulation in Response to Very-Low and High CO2in Microchloropsis gaditanaNIES2587. Frontiers in Plant Science 11: 981.
8.Billey E,et al. 2021. Characterization of theBubblegum acyl-CoA synthetase of Microchloropsisgaditana. Plant Physiology 185(3): 815-835.
9.Vonshak A,et al. 2020. Photosyntheticcharacterization of two Nannochloropsisspecies and its relevance to outdoor cultivation. Journal of Applied Phycology32(2):909-922.
10.Dienst D,et al. (2020). High density cultivationfor efficient sesquiterpenoid biosynthesis in Synechocystissp. PCC 6803. Scientific Reports 10(1): 5932.
11.Weiner I, et al. 2020. CSO -A sequenceoptimization software for engineering chloroplast expression in Chlamydomonas reinhardtii. AlgalResearch 46: 101788.
12.Akma C, et al. 2020. Two-phase method ofcultivating Coelastrellaspecies forincreased production of lipids and carotenoids. Bioresource Technology Reports9: 100366.
13.Cecchin M, et al. 2020. Improved lipid productivityin Nannochloropsisgaditana innitrogen-replete conditions by selection of pale green mutants. Biotechnologyfor Biofuels 13(1): 78.
14.Alvarenga D, et al. 2020. AcnSP – A Novel SmallProtein Regulator of Aconitase Activity in the Cyanobacterium Synechocystissp. PCC 6803. Frontiers inMicrobiology 11: 1445.
15.Zhang B, et al. 2020. The carbonate concentrationmechanism of Pyropia yezoensis(Rhodophyta): evidence from transcriptomics and biochemical data. BMC PlantBiology 20(1): 424.
16.Nzayisenga, JC,et al. 2020. Effects of light intensityon growth and lipid production in microalgae grown in wastewater. Biotechnologyfor Biofuels 13(284): 1179-1184.
17.Cecchin M,et al. 2020. Improved lipidproductivity in Nannochloropsis gaditanain nitrogen-replete conditions by selection of pale green mutants.Biotechnology for Biofuels 13(6): 312.
18.Flamholz AI,et al. 2020. Functional reconstitutionof a bacterial CO2concentrating mechanism in Escherichia coli. eLife 9: e59882.
19.Gupta JK, et al. 2020. Overexpression ofbicarbonate transporters in the marine cyanobacterium Synechococcussp. PCC 7002 increases growth rate and glycogenaccumulation. Biotechnology for Biofuels 13: 17.
20.Valev D,et al. 2020. Testing the Potential ofRegulatory Sigma Factor Mutants for Wastewater Purification or Bioreactor Runin High Light. Current Microbiology 77(8) : 1590-1599.
21.Yao L,et al.. 2020. Pooled CRISPRi screeningof the cyanobacterium Synechocystissp PCC 6803 for enhanced industrial phenotypes. Nature Communications 11(1):1666.
22.Shrameeta S, et al. 2020. Glycogen MetabolismSupports Photosynthesis Start through the Oxidative Pentose Phosphate Pathwayin Cyanobacteria1. Plant Physiology 182(1):507-517.
23.AlessandraB,et al. 2020. PhotosynthesisRegulation in Response to Fluctuating Light in the Secondary Endosymbiont Alga Nannochloropsis gaditana. Plant &Cell Physiology 61(1): 41-52..